BD intensifies syringe output to meet U.S. demand amid China quality concerns

BD’s North Canaan plant has increased its output of syringes by more than 40% to meet the needs of U.S. healthcare customers after the FDA launched an ongoing investigation into quality issues with plastic syringes made in China.
Provided


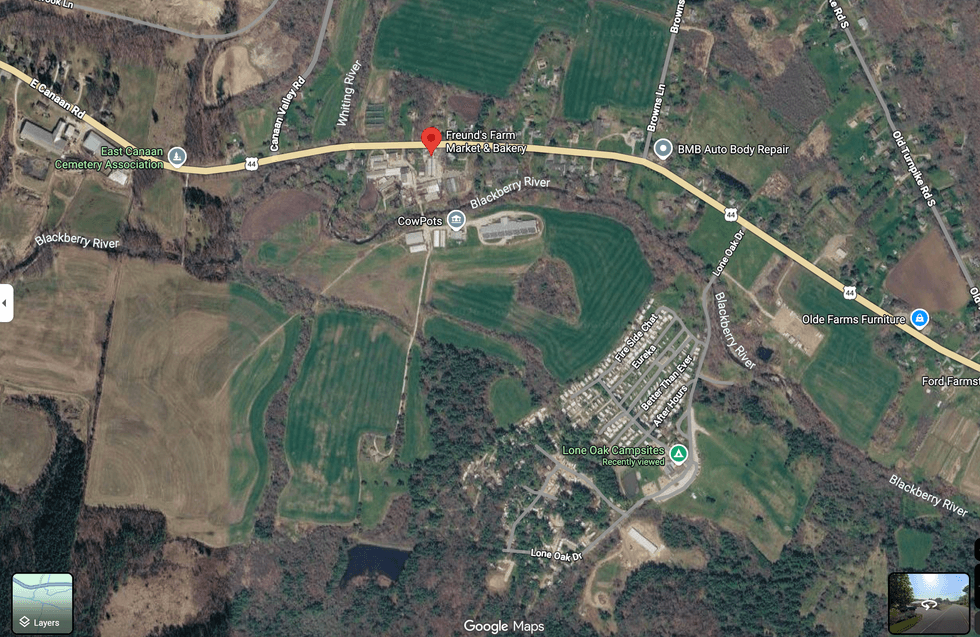 The drowning occurred in a private pond behind Freund's Farm Market and Bakery in North Canaan, west of the Lone Oak Campsites.
The drowning occurred in a private pond behind Freund's Farm Market and Bakery in North Canaan, west of the Lone Oak Campsites.